Which of these have physical and chemical properties resembling calcium ?Ĭompare and contrast the arrangement of elements in Mendeleev’s Periodic Table and the Modern Periodic Table. In the Modern Periodic Table, calcium (atomic number 20) is surrounded by elements with atomic number 12, 19, 21 and 38. How does the electronic configuration of an atom relate to its position in the Modern Periodic Table ? It has an atomic number 7 and an atomic weight of 14.0067 FIGURE 2.3 Lewis dot. (d) Which type of ion, cation or anion, will be formed by element A ? Each bond in a triple bond is a pair of electrons, so a triple bond. (c) Will C be larger or smaller in size than B ? (b) State whether C is more reactive or less reactive than A. The most abundant isotope (as they are called) is ''14N, however, there exist isotopes where the nitrogen nucleus contains more than 7 neutrons. If a nucleus possesses 7 protons, 7 fundamental positive particles, it is by definition a nitrogen atom. (a) State whether A is a metal or non-metal. Because there exist nuclei with with 7 protons and MORE OR LESS than 7 neutrons that is nitrogen possesses a number of ISOTOPES. The positions of three elements A, B and C in the periodic table are shown below : (b) To which of the following elements would it be chemically similar ? (Atomic numbers are given in parentheses.) (a) What is the atomic number of this element ? as fluorine have in common ?Īn atom has electronic configuration 2, 8, 7. (b) What property do all elements in the same column of the Periodic Table. (a)What property do all elements in the same column of the Periodic Table as boron have in common ? 
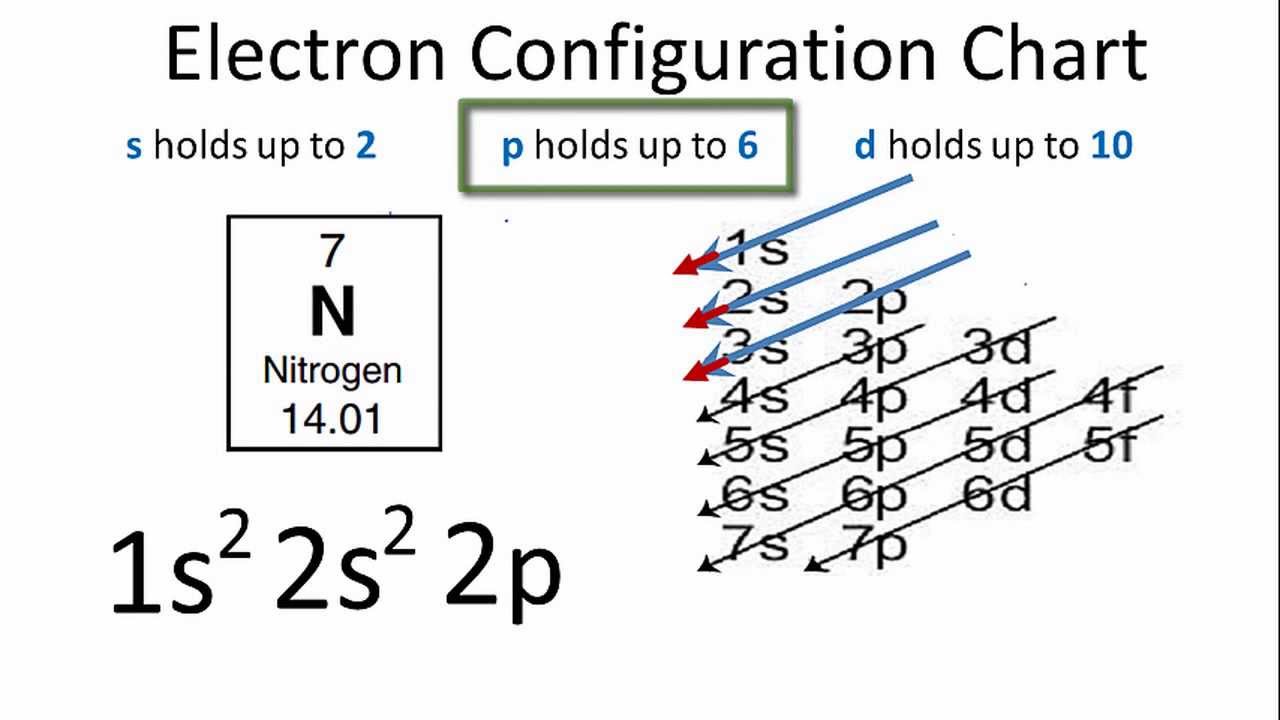
(e)twice as many electrons in its second shell as in its first shell ? (d) a total of two shells with three electrons in its valence shell. (c) a total of three shells, with four electrons in its valence shell ? (b) the electronic configuration 2, 8, 2 ? (a) two shells, both of which are completely filled with electrons ? X would most likely to be in the same group of the periodic table as (c)The atoms lose their electrons more easily.Įlement X forms a chloride with the formula \( XCl_2, \) which is solid with a high melting point. (b) The number of valence electrons increases. Since the 1s orbital can only hold two electrons, the next two electrons for N go into the 2s. (a) The elements become less metallic in nature. The mass number of nitrogen is 14, and its atomic number is 7. Which of the following statements is not a correct statement about the trends wlien going from left to right across the periods of Periodic Table. NCERT solutions of related questions for Periodic Classification of Elements 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
